Contact:
Heather Williams
+ 44 (0) 7557 001205
Heather_Williams@europe.bd.com
BD Launches the PAXgene® Blood DNA Tube in Europe
Oxford, United Kingdom (January 28, 2014) – BD (Becton, Dickinson and Company), a leading global medical technology company, today announced the commercial availability of the CE-marked PAXgene® Blood DNA Tube in Europe. The PAXgene® Blood DNA Tube is the first blood collection tube designed specifically for in vitro diagnostic (IVD) genomic DNA testing. The product was developed by PreAnalytiX GmbH, a joint venture between BD and QIAGEN.
DNA testing has advanced significantly since the publication of the first draft of the human genome in 2000. While swab and saliva samples are popular, blood remains the sample of choice for clinical diagnostic testing and identification of new biomarkers for disease research.
To improve the collection of DNA in these applications, the PAXgene® Blood DNA Tube performance has been comprehensively analysed. “The validated performance data we provide to clinical scientists for the PAXgene® Blood DNA Tube should reduce the chances of preanalytical error, ensuring the best quality sample for accurate IVD test performance and for blood DNA storage related to the identification of new biomarkers of disease or drug actions”, comments Frank Augello, General Manager, PreAnalytiX GmbH.
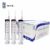
The PAXgene® Blood DNA Tube is a 2.5 ml blood collection tube that stabilizes genomic DNA with validated time and temperature parameters. The tube incorporates workflow efficiency features including a 2D bar code on the label and a tube closure that clearly identifies this unique DNA tube.
The PAXgene® Blood DNA Tube is a constituent of a complete molecular diagnostic portfolio of PreAnalytiX GmbH products comprising DNA isolation kits, automation and analysis equipment. The initial target customers for the PAXgene® Blood DNA Tube are those requiring validated high quality blood DNA - blood DNA testing laboratories and biorepositories.
For more information, contact your local BD customer services office (www.bd.com) or visit http://www.preanalytix.com/product-catalog/blood/dna/products/paxgene-blood-dna-tube-ce/.
About PreAnalytiX GmbH
PreAnalytiX GmbH, a joint venture between BD and QIAGEN, develops, manufactures and sells integrated and standardized systems for collection, stabilization and purification of RNA, microRNA and DNA from blood, bone marrow and tissue specimens. The Company serves healthcare institutions, academic researchers, clinical laboratories and the pharmaceutical industry with a broad array of manual and automated products.
For more information, please visit www.preanalytix.com
About BD
BD is a leading medical technology company that partners with customers and stakeholders to address many of the world’s most pressing and evolving health needs. Our innovative solutions are focused on improving drug delivery, enhancing the diagnosis of infectious diseases and cancers, supporting the management of diabetes and advancing cellular research. We are nearly 30,000 associates in 50 countries who strive to fulfill our purpose of “Helping all people live healthy lives” by advancing the quality, accessibility, safety and affordability of healthcare around the world. For more information, please visit www.bd.com.
About Qiagen
QIAGEN N.V. is a Netherlands holding company publically traded on NASDAQ and Frankfurt Prime Standard. The Company is the leading global provider of Sample & Assay Technologies that are used to transform biological materials into valuable molecular insights. Sample technologies are used to isolate and process DNA, RNA and proteins from biological samples such as blood or tissue. Assay technologies are then used to make these isolated biomolecules visible and ready for interpretation. QIAGEN markets more than 500 products around the world, selling both consumable kits and automation systems to customers through four customer classes: Molecular Diagnostics (human healthcare), Applied Testing (forensics, veterinary testing and food safety), Pharma (pharmaceutical and biotechnology companies) and Academia (life sciences research). As of September 30, 2013, QIAGEN employed more than 4,100 people in over 35 locations worldwide. Further information can be found at http://www.qiagen.com.
###




















 熱線訂購:
熱線訂購: 手機(jī)/微信:
手機(jī)/微信: